Mysteries of Water Ice at Extreme Temperature and Pressures
10 Aug 2023When pure natural water is cooled below freezing point, it transform to a tetrahedral arrangement and becomes crystaline solid. At this point we call it as an ice structure. This phenomena is called a phase transition. The structure after phase transition depends on thermodynamic conditions such as temperature, pressure, and chemical purity. For instance, at low temperature and/or high pressure, water is found to transform into other forms of crystaline structures. Some of these structures are not possible to be seen under earthly conditions. These different ice structures of water is possible through water molecule’s unique structure and its ability to form hydrogen bonds. The hydrogen bonding of water at different regieme of the phase diagram is the reason why water is an extremely challenging molecule to understand.
The concept of the hydrogen bond, as proposed by Linus Pauling, plays a crucial role in the formation of H2O ice structures. A hydrogen bond can be represented as O–H⋯O, where the hydrogen atom is connected to the donor oxygen atom through a short covalent bond and to the acceptor through a longer bond.
What makes the structures of ice varied is the ability of a water molecule to form four hydrogen bonds, despite having just three atoms. This means two atoms can serve as hydrogen donors and two as hydrogen acceptors. Unlike in water clusters or liquid water, all four hydrogen bonds are fully formed in ice, leading to a tetrahedral coordination around each water molecule. This specific local coordination pattern is referred to as the Walrafen pentamer and is found in almost all ice structures
 Fig: Walrafen pentamer [Ref: Brewer, Peter G., et al. “The speciation of water in sea water and in gelatinous marine animals.” _Marine Chemistry_ 195 (2017): 94-104.]
Fig: Walrafen pentamer [Ref: Brewer, Peter G., et al. “The speciation of water in sea water and in gelatinous marine animals.” _Marine Chemistry_ 195 (2017): 94-104.]
Most of the ice structures were predicted in theory and simulation works. Some of the structures were experimentally confirmed in later years. These prediction consists of water structures that were obtained at different temperarture and pressure.
Let’s look at the phase diagram of water at an intermediate temperature and pressure regime.
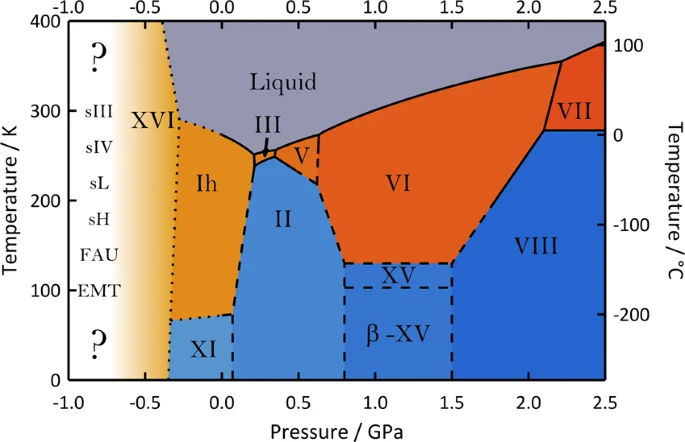
It consists of so many water ice structures. Infact according to Engel and Ceriotti there are total 51 ice polymorphs and more than 30 of these are still not yet reported in experiments. The above phase diagram also consists of negative pressure. Ice structures of significantly less density than common heagonal ice are found in this negative pressure regime. These have open structures that are large enough to cage a guest molecule similar to clathrate hydrates. Such open ice forms without any caged molecule inside are supposed to transform to hexagonal ice under tensile stress. In the past, experiments with such ice structure were mostly unsuccessful because the icicles rupture easily. However, experirment on liquid water, done at negative pressure and avoiding cavitation, we able to reach a stable vapour phase. Ice XVI and XVII were obtained experimentally. These are empty clathrate hydrates which should be stable only at negative pressure, and would soon transform to ice Ih as soon as the pressure becomes positive (from the above phase diagram). Apart from these two, computer simulation have predicted a number of stable structuures at negative pressures which are inspired from known guest-filled clathrate hydrates or zeolite type networks (listed in the negative pressure region). Experimentally, empty hydrogen-bond network of these predicted ice structures are yet to be observed. Some of them could also be metastable states.
What are the ice structures at high pressure? At pressure exceeding 50 GPa, the Walrafen pentamer breaks down. In this high pressure regime the hydrogen atom centers itself between two oxygen atoms. The molecular nature of water slowly breaks to a point wherer we cannot assign hydrogens to any particular oxygen atom. Here, the H atoms jump back and forth between the two O atoms (labelled dynamic VII/X in the phase diagram below).
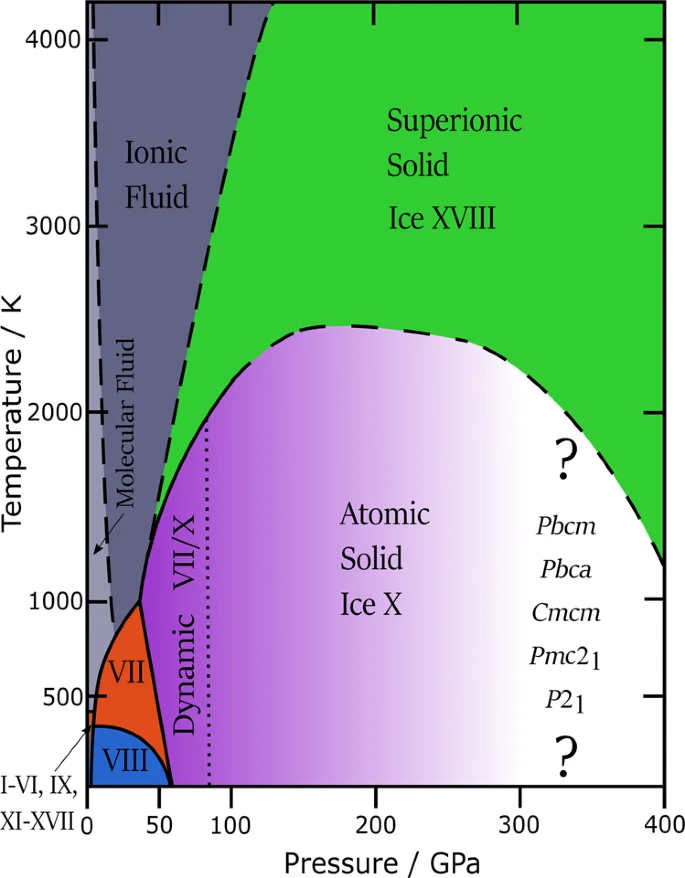
Crorssing 100 GPa and below 2500 K, enen the hydrogen atoms get frozen at the centre of the two oxygen atoms (labelled Ice X). Experimentally Ice X was observed in 1984. Astonishingly, this ice does not melt even beyond 2500 K. It undergoes a dynamic transition instead. This transition makes the Hydrogen atoms become mobile in the lattice of Oxygen atoms which remains fixed in their positions. This is known as superionicity.
This kind of ice was first predicted in 1988 by researchers at the University of Pennsylvania through Molecular Dynamics (MD) simulations. However, experirmental evidence of solid superionic ice XVIII was observed very recently (2019) in X-ray experiments on laser-shocked water. As the hydrogen atoms are mobile, Ice XVIII is very much conductive like metals. The delocalized hydrogen atoms in ice XVIII plays the same role as delocalized electrons in metals. This hypothesis was confirmed when it was observed that the optical reflectivity remained low (which relates to low electronic conductivity (Hagens-Ruben equation)) even as the electrical conductivity shot up ($\sigma \sim 100 S cm^{–1}$), ruling out free electrons and thereby showing the conductivity must arise from delocalised protons. The interior of Neptune and Uranus may contain a significant amount of superionic ice See here. This theory might offer some clarification for the enigmatic magnetic fields of Neptune and Uranus, which the Voyager 2 probe discovered to be very different from the straightforward dipolar fields on Earth and other planets.
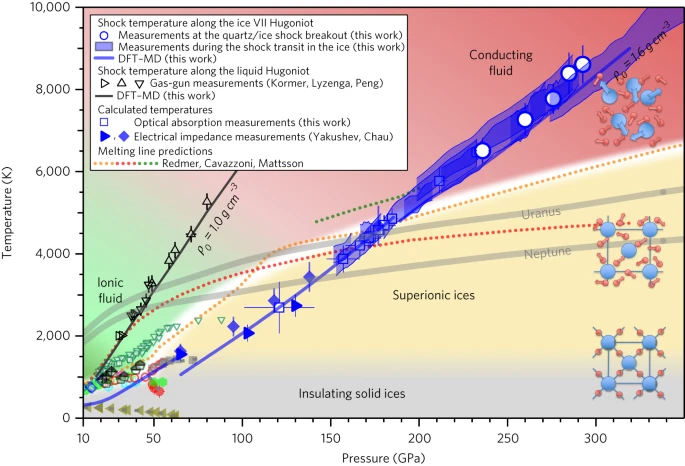 Ref: H2O phase diagram at planetary interior conditions
Ref: H2O phase diagram at planetary interior conditions
For our day to day activity, it is almost impossible to see anything more than hexagonal Ice Ih. It is the only form relevant to the Earth’s environmental conditions, as even the thickest ice slabs do not provide the necessary pressure to pass it into high-pressure polymorphs. Cubic ice, ice Ic, recently isolated in pure form, plays a role at very low temperatures in the higher atmosphere in the nucleation of ice; ice VI, the lowest high-pressure ice phase at room temperature has been spectroscopically found in diamond inclusions, and ice VII has been prerdicited to be present on Earth in cold subduction zones.
The search for ice phases continues, and the following articles could be a good read: